Unified Platform for Electronic-Sourced Digital Biomarkers in CNS Trials
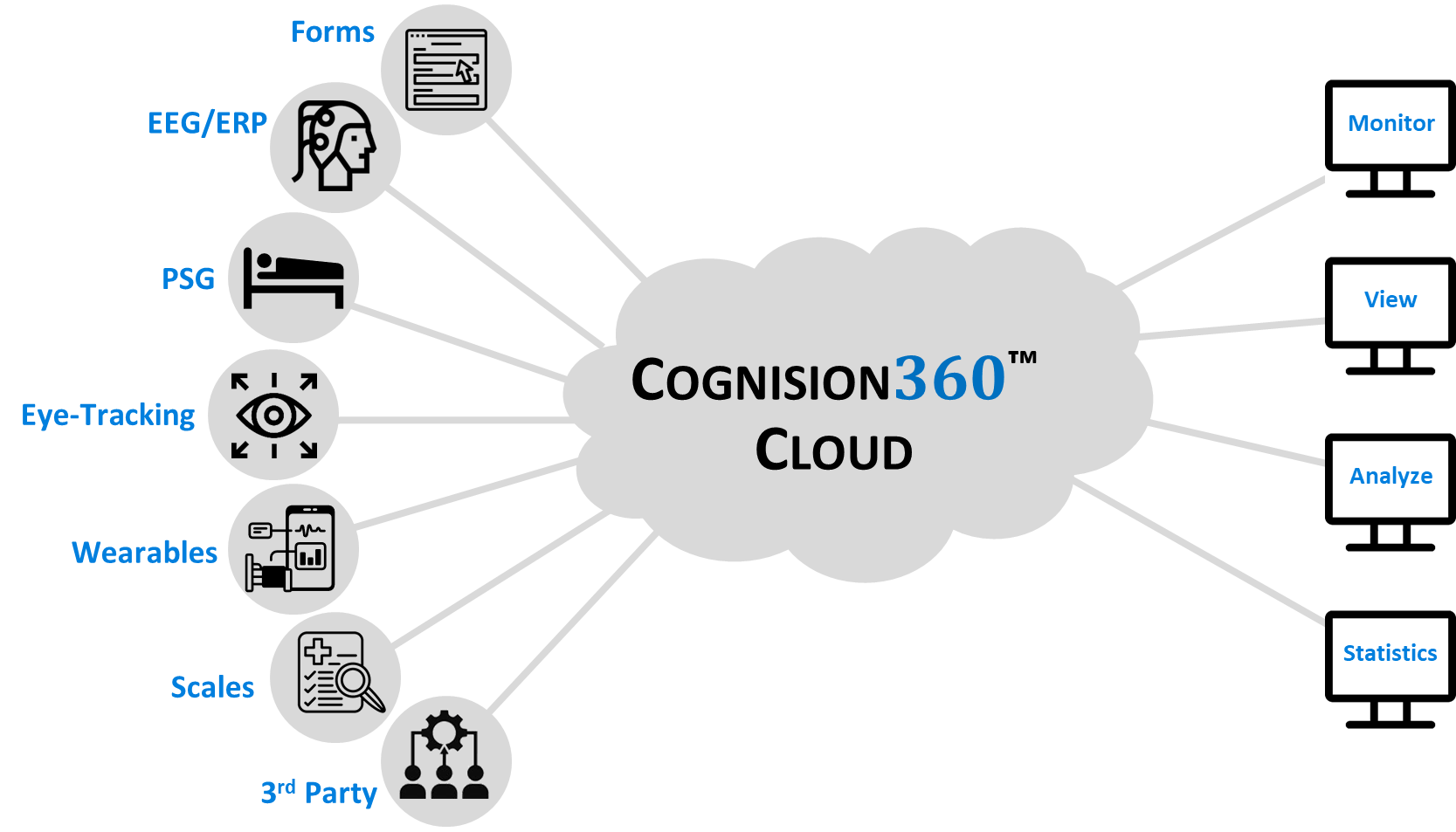
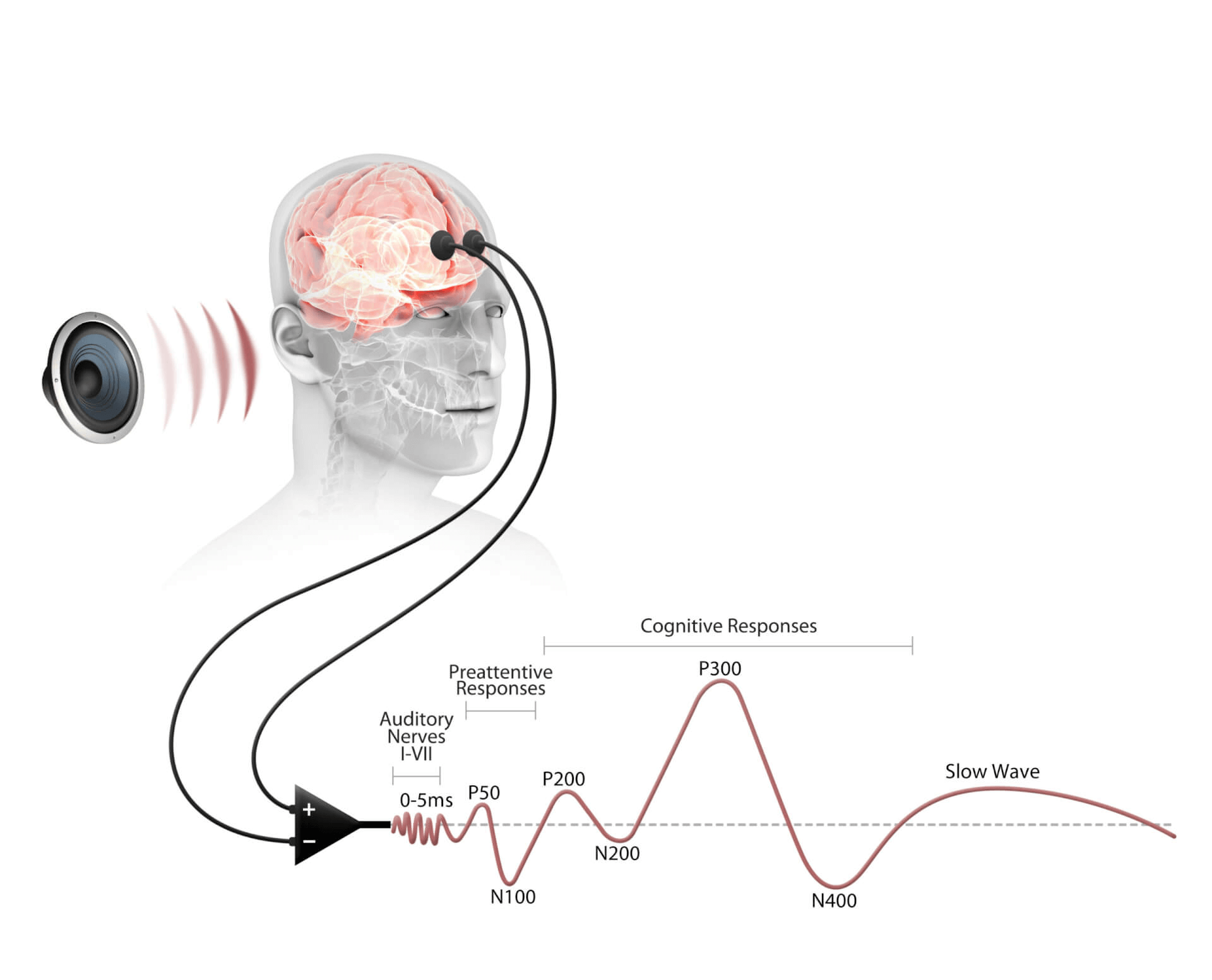
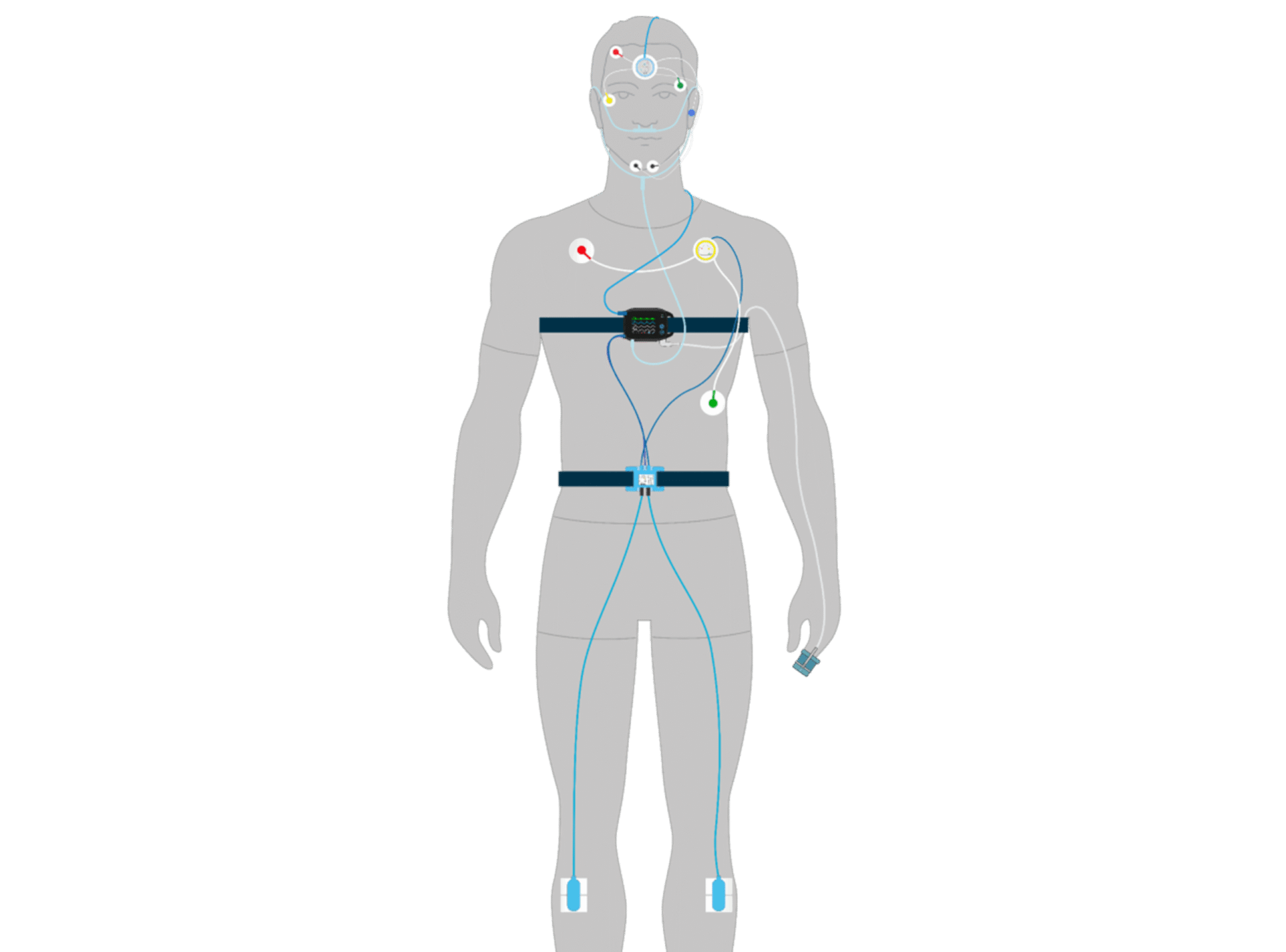
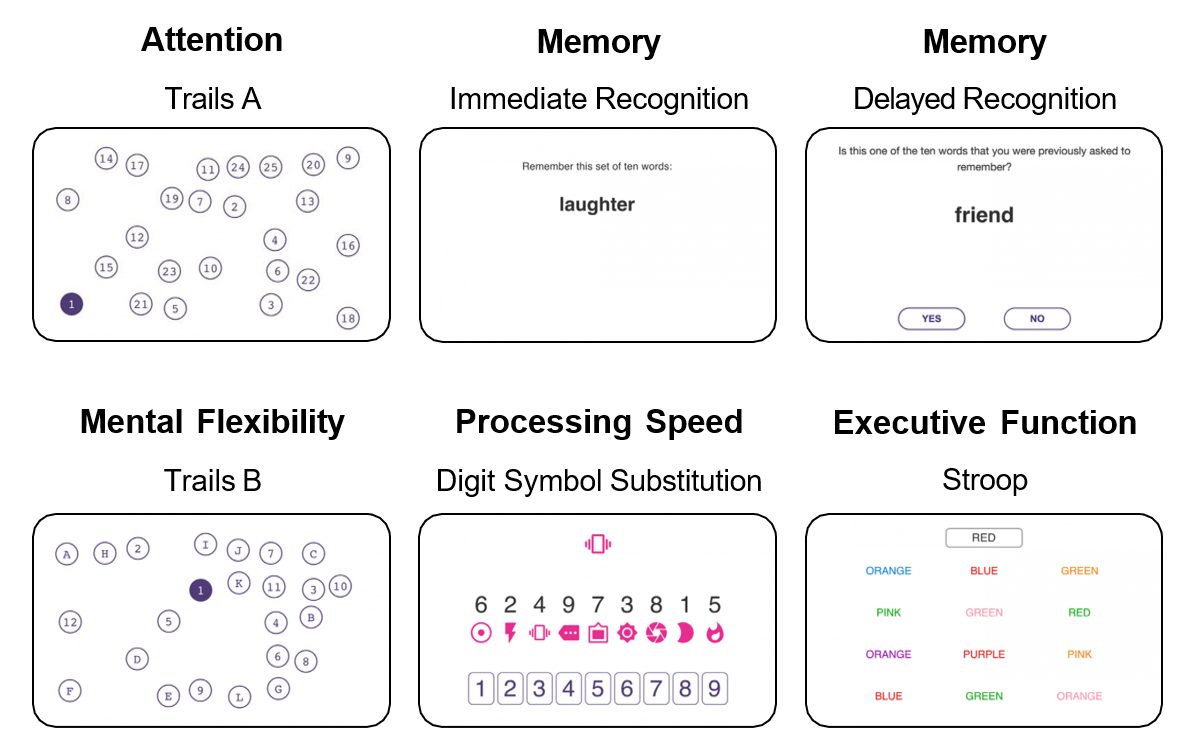
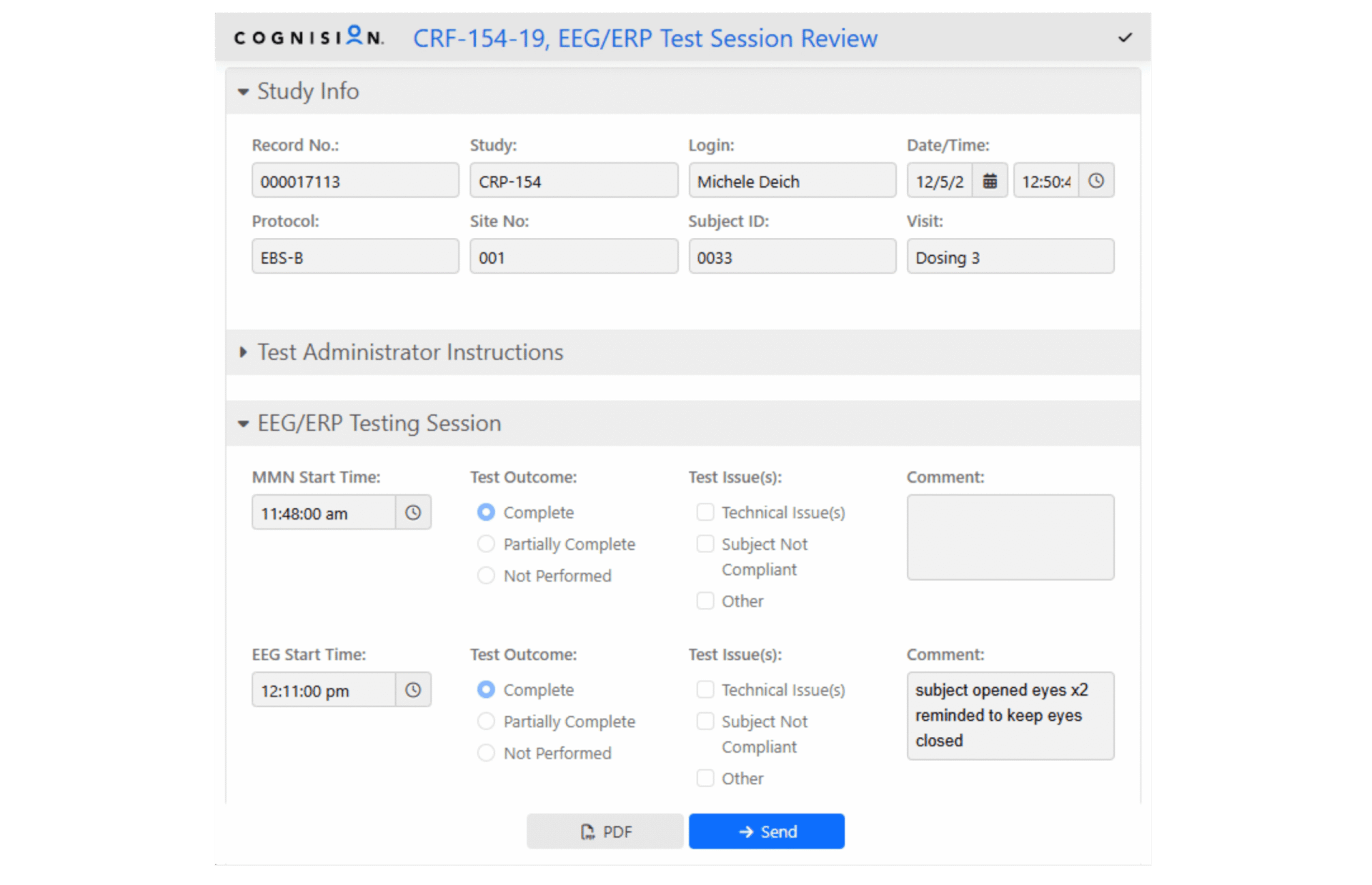
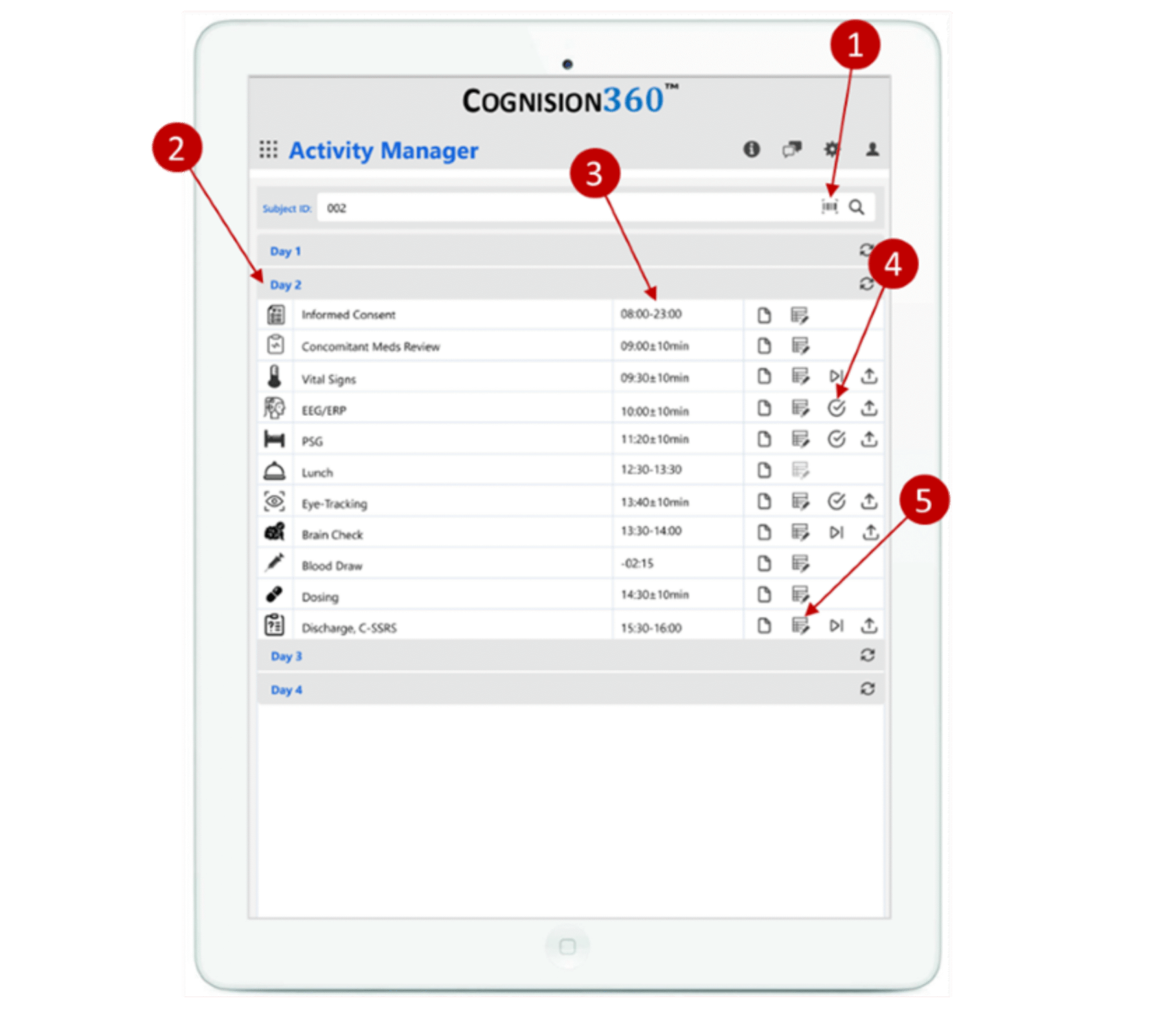
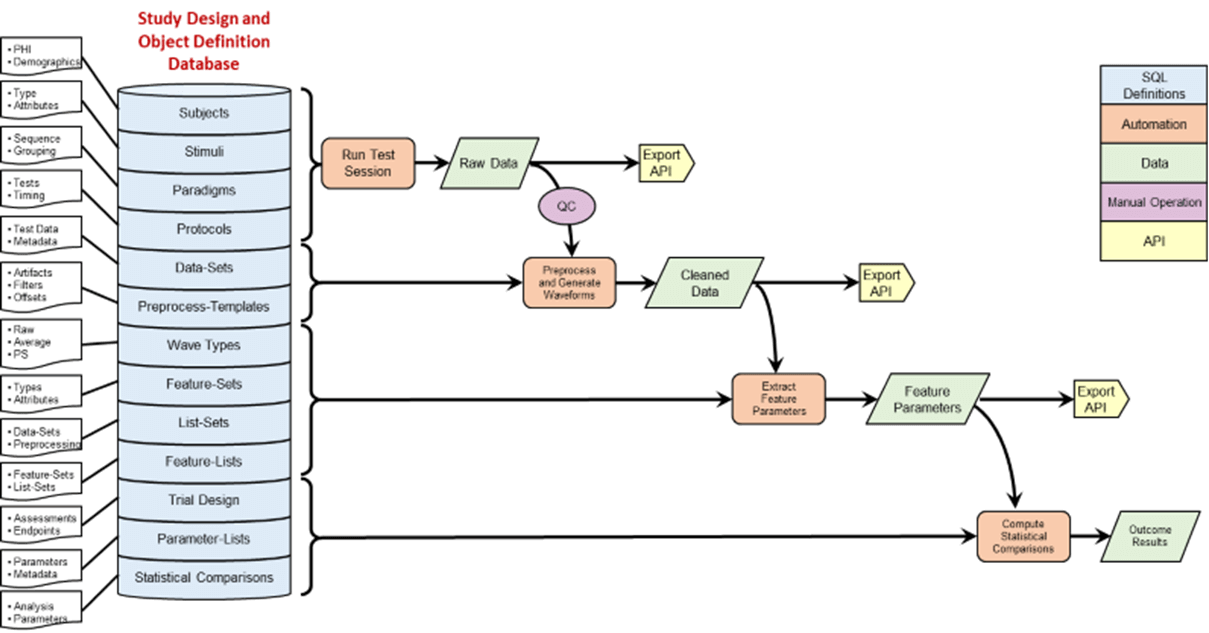
Cognision360™ streamlines digital biomarker implementation in central nervous system clinical trials. The platform integrates data collection, management, and analysis from multiple sources—advanced instrumentation, wearable sensors, mobile assessments, and remote monitoring tools—into a single unified system. Researchers gain real-time, objective measurements of cortical function, cognition, motor performance, sleep patterns, and behavioral changes, all captured electronically and managed through one interface.